
Not an actual patient.
Efficacy that lasts1
Just one dose every 3 months1
The dosing schedule aligns with American Association of Clinical Endocrinology (AACE) recommendations for triglyceride level testing—once every 3 months, or more frequently when necessary.1,2,*
As an adjunct to diet,
Redemplo® is self-administered as a single subcutaneous 25-mg injection every 3 months1
If a dose is missed, Redemplo should be administered as soon as possible, and regular dosing should resume 3 months after administered dose.1
Redemplo should be administered in any of the following locations3:

Upper Arm
(when administered by caregiver)
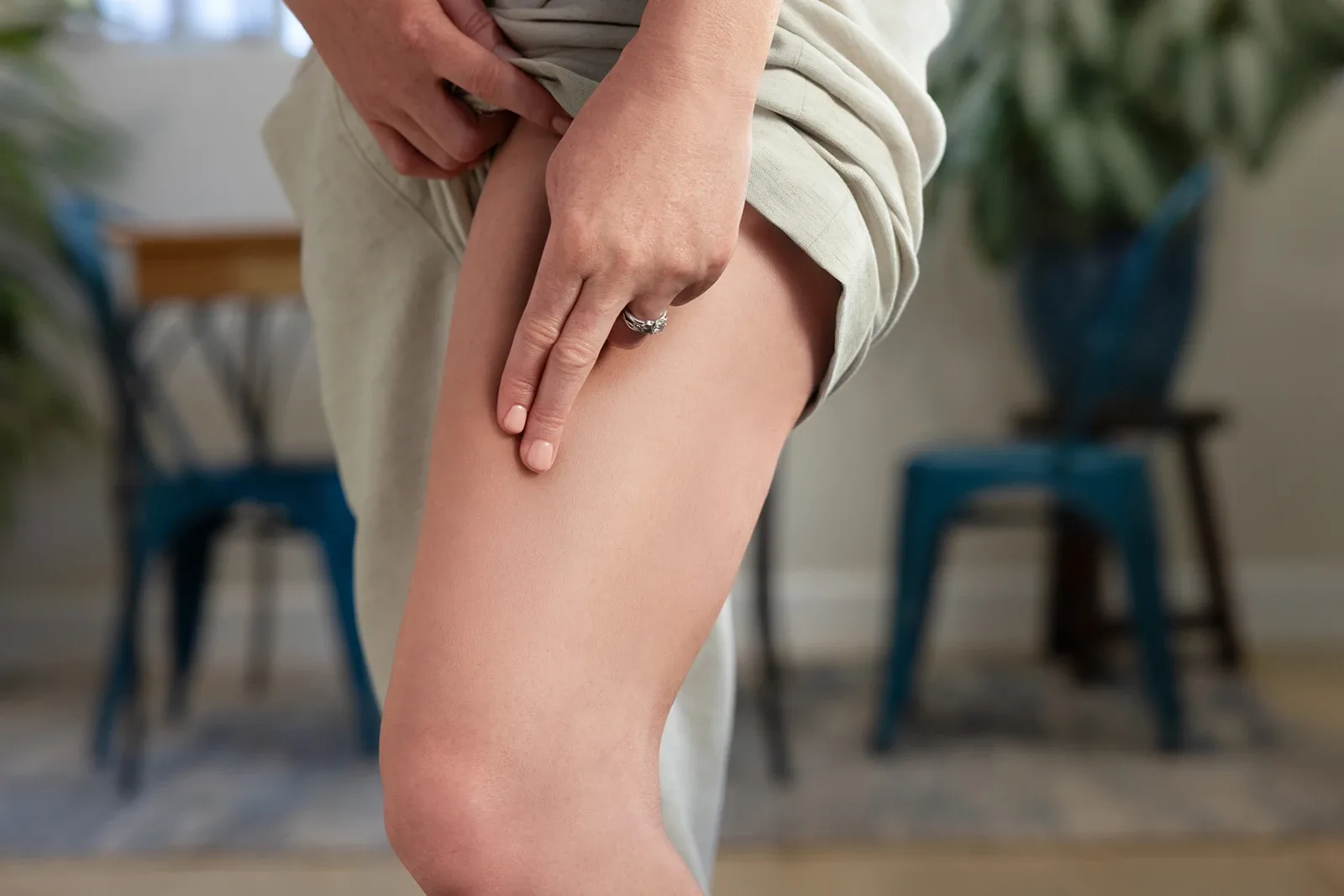
Thigh

Abdomen
How to administer Redemplo
This video is a helpful resource for your patients with FCS and their caregivers on how to properly administer Redemplo.
Are you ready to help your patients GO LO with Redemplo?

Not an actual patient.
Proven results in patients with FCS1

Not an actual patient.
Resources and support are available
FCS, Familial Chylomicronemia Syndrome.
REFERENCES Collapse
- Redemplo. Prescribing information. Arrowhead Pharmaceuticals, Inc.; 2025.
- Handelsman Y, Jellinger PS, Guerin CK, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the management of dyslipidemia and prevention of cardiovascular disease algorithm – 2020 executive summary. Endocr Pract. 2020;26(10):1196-1224.
- Redemplo. Instructions for use. Arrowhead Pharmaceuticals, Inc.; 2025.
Indication & Important Safety Information
Indication
REDEMPLO® (plozasiran) is indicated as an adjunct to diet to reduce triglycerides in adults with familial chylomicronemia syndrome (FCS).
Important Safety Information
CONTRAINDICATIONS: None.
ADVERSE REACTIONS: Most common adverse reactions in REDEMPLO treated patients (incidence ≥10% of patients treated with REDEMPLO and > 5% more frequently than with placebo) are hyperglycemia, headache, nausea, and injection site reaction.