
Not an actual patient.
Redemplo® demonstrated a well-tolerated safety profile1,2
Redemplo safety
Adverse Reactions Occurring in ≥10% of Redemplo-Treated Patients and >5% More Frequently Than With Placebo1
| Adverse Reactions | Placebo (N=25) (%) | Redemplo* (N=50) (%) |
|---|---|---|
| Hyperglycemia† | 2 (8) | 10 (20) |
| Headache | 2 (8) | 8 (16) |
| Nausea | 2 (8) | 7 (14) |
| Injection site reaction† | 1 (4) | 5 (10) |
Redemplo has no contraindications, warnings, or precautions in its FDA-approved label.1
*Patients were treated with either 25 mg or 50 mg of plozasiran. Plozasiran 50 mg
is not an approved dosing regimen for FCS.1
†Grouped terms composed of several similar terms.1
Low rates of discontinuation1,3
Rates of discontinuation due to adverse reactions
6% of patients receiving Redemplo discontinued treatment due to hyperglycemia or urticaria vs 0% receiving placebo1
Rates of discontinuation due to acute pancreatitis
No patients taking Redemplo in PALISADE discontinued due to acute pancreatitis vs 12% receiving placebo2
PALISADE STUDY DESIGN Collapse
Redemplo is the only FDA-approved treatment studied in both genetically confirmed and clinically diagnosed patients living with FCS1,2
The PALISADE Study was a pivotal, randomized, placebo-controlled, double-blind trial in adult patients with genetically confirmed or clinically diagnosed FCS maintained on a low-fat diet (≤20 grams of fat per day), where 75 patients were randomized to receive 4 total doses of Redemplo or a matching placebo subcutaneously once every 3 months.1
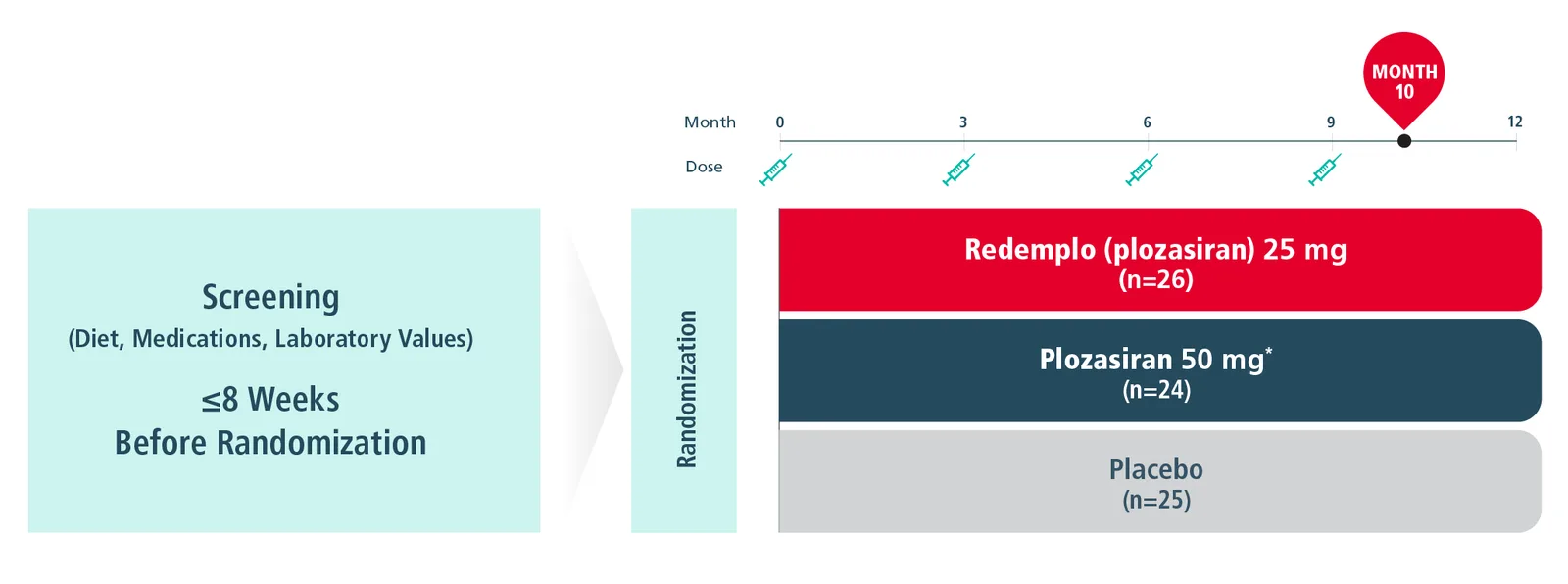
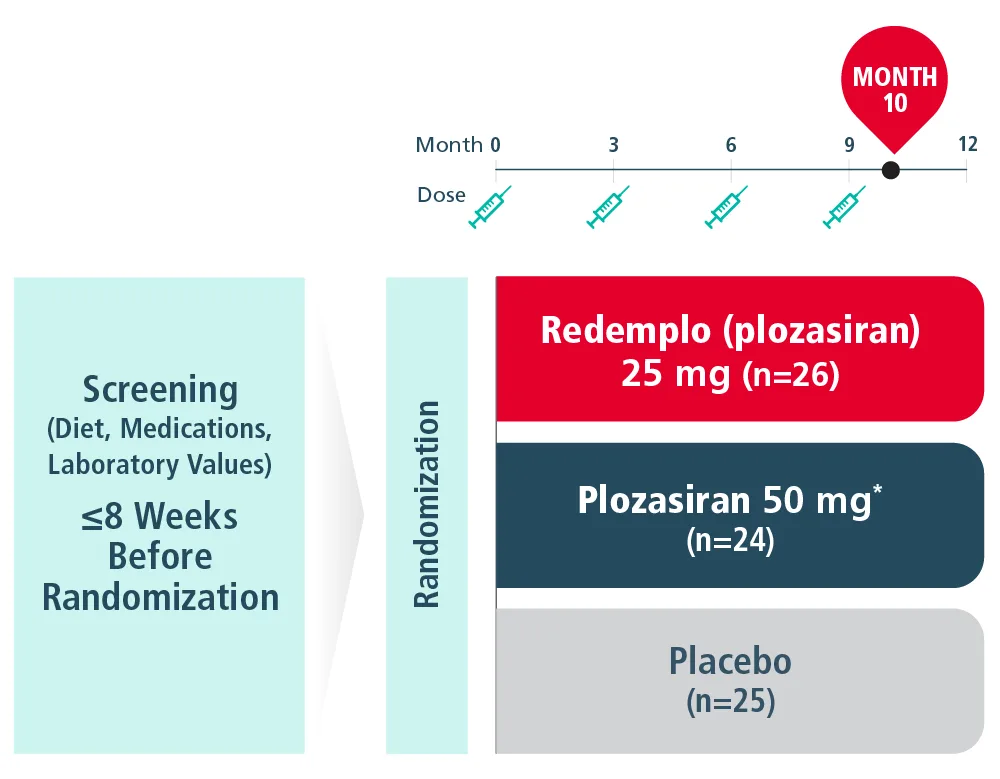
Primary Endpoint1
- Median percent change from baseline in the fasting triglycerides level at Month 10
Multiplicity-Controlled Key Secondary Endpoints1
- Percent change from baseline at Months 10 and 12 (averaged) in fasting triglycerides
- Percent change from baseline at Month 10 in fasting apoC-III
- Percent change from baseline at Month 12 in fasting apoC-III
- Incidence of positively adjudicated events of acute pancreatitis during the randomized period
- Evidence of low lipoprotein lipase activity
*Plozasiran 25-mg and 50-mg doses resulted in similar triglyceride reductions. Plozasiran 50 mg is not an approved dosing regimen for FCS.1
KEY INCLUSION AND EXCLUSION CRITERIA Collapse
Key Inclusion and Exclusion Criteria
Adult patients with FCS who were clinically diagnosed or genetically confirmed were evaluated in PALISADE. Inclusion criteria included fasting triglyceride levels of ≥880 mg/dL that were unresponsive to traditional triglyceride-lowering therapies in patients with at least 3 triglyceride measurements >1000 mg/dL and at least ONE of the following1,2:
- Recurrent episodes of acute pancreatitis not caused by alcohol or cholelithiasis OR
- Hospitalizations for severe abdominal pain without other explainable cause OR
- History of childhood pancreatitis OR
- Family history of HTG-induced acute pancreatitis OR
- Prior genetic testing diagnostic of FCS
Exclusion criteria included uncontrolled diabetes, use of corticosteroids or anabolic steroids, and chronic kidney disease.2
BASELINE CHARACTERISTICS Collapse
| Redemplo (n=26) | Placebo (n=25) | |
|---|---|---|
| Median fasting triglycerides at baseline | 2008 mg/dL | 2053 mg/dL |
| Clinically diagnosed | 54% | 44% |
| Genetically confirmed | 46% | 56% |
| Diabetes | 15% | 32% |
| History of pancreatitis in the prior 5 years | 54% | 68% |
Across treatment groups, the mean age was 46 years and 49% of patients were male. Seventy-three percent (73%) of patients were White, 21% were Asian, and 5% were reported as other races; 3% identified as Hispanic or Latino ethnicity.1-3
Rapid and sustained apoC-III reduction1

Not an actual patient.
Proven results in patients with FCS1
apoC-III, apolipoprotein C-III; FCS, Familial Chylomicronemia Syndrome; HTG, hypertriglyceridemia.
REFERENCES Collapse
- Redemplo. Prescribing information. Arrowhead Pharmaceuticals, Inc.; 2025.
- Watts GF, Rosenson RS, Hegele RA, et al. Plozasiran for managing persistent chylomicronemia and pancreatitis risk. New Engl J Med. 2025;392(2):127-137.
- Data on file. Arrowhead Pharmaceuticals, Inc.; 2024.
Indication & Important Safety Information
Indication
REDEMPLO® (plozasiran) is indicated as an adjunct to diet to reduce triglycerides in adults with familial chylomicronemia syndrome (FCS).
Important Safety Information
CONTRAINDICATIONS: None.
ADVERSE REACTIONS: Most common adverse reactions in REDEMPLO treated patients (incidence ≥10% of patients treated with REDEMPLO and > 5% more frequently than with placebo) are hyperglycemia, headache, nausea, and injection site reaction.