
Not actual patients.
How Redemplo® helps your patients GO LO
Redemplo is the only siRNA therapy that is designed to silence the mRNA for apoC-III, an important target for reducing triglycerides in patients with FCS1-3
Redemplo uses RNA interference to selectively degrade apoC-III mRNA in hepatocytes—this reduces hepatic and serum apoC-III protein levels, enhancing triglyceride breakdown and promoting clearance of triglyceride-rich lipoprotein remnants—ultimately lowering serum triglycerides.1
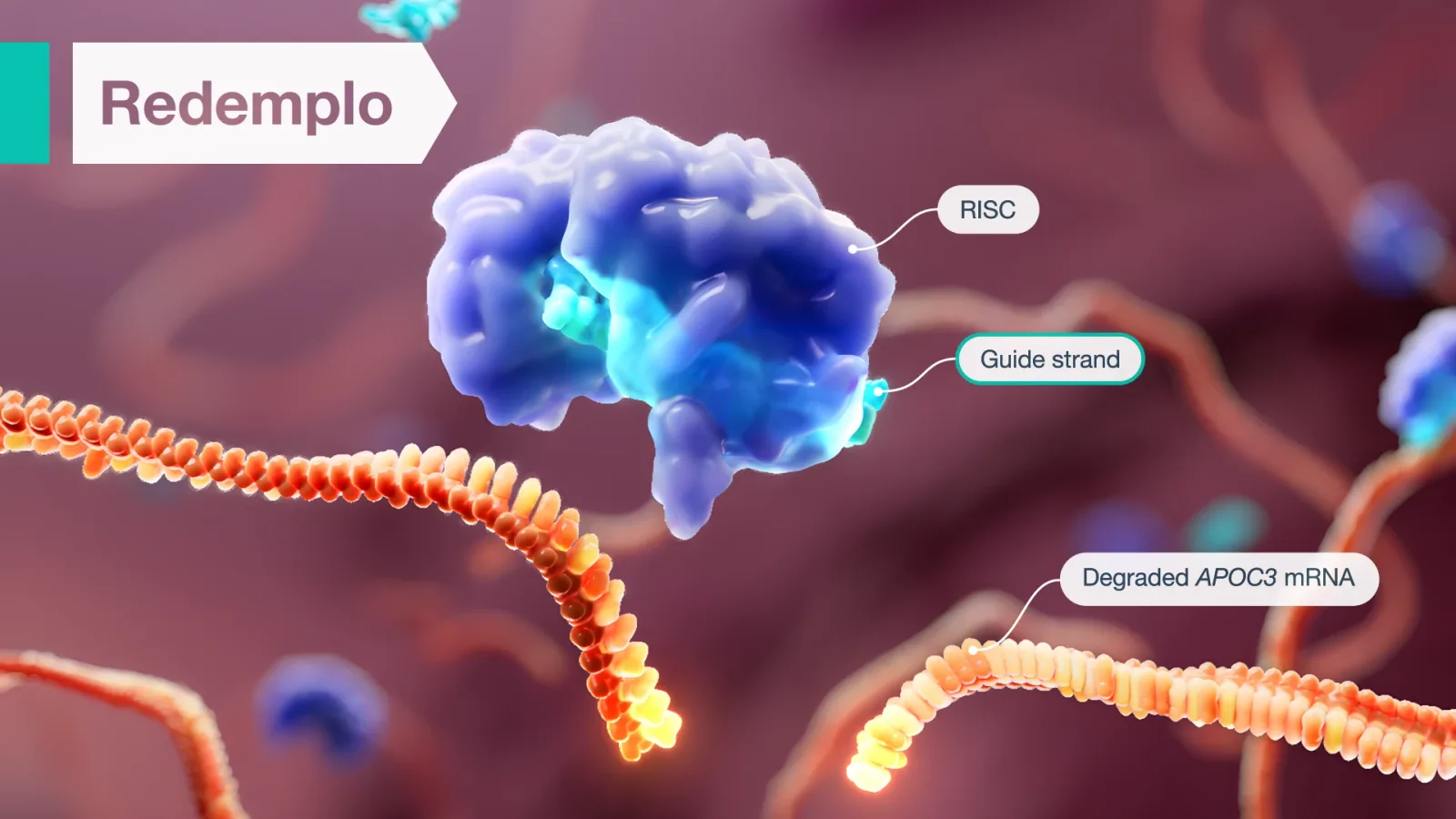
See how Redemplo works
Redemplo resulted in a rapid and sustained reduction in apoC-III levels4
Secondary Endpoint: apoC-Ill Levels Were Reduced as Early as Month 1 and Sustained Through Month 10 with Redemplo 25 mg5
Median apoC-III values at baseline were 39 mg/dL for Redemplo (n=26) and 39 mg/dL for placebo (n=25).5
By reducing apoC-III levels, Redemplo helps to lower triglyceride levels in adults with FCS1,4
Convenient dosing—one dose every 3 months4

Not actual patients.
Is Redemplo right for your patients?
apoC-III, apolipoprotein C-III; FCS, Familial Chylomicronemia Syndrome; mRNA, messenger RNA; RNA, ribonucleic acid; siRNA, small interfering RNA.
REFERENCES Collapse
- Data on file. Arrowhead Pharmaceuticals, Inc.; 2024.
- Gaudet D, Pall D, Watts GF, et al. Plozasiran (ARO-APOC3) for severe hypertriglyceridemia: the SHASTA-2 randomized clinical trial. JAMA Cardiol. 2024;9(7):620-630. doi:10.1001/jamacardio.2024.0959
- Gaudet D, Clifton P, Sullivan D, et al. RNA interference therapy targeting apolipoprotein C-III in hypertriglyceridemia. NEJM Evid. 2023;2(12):EVIDoa2200325. doi:10.1056/EVIDoa2200325
- Redemplo. Prescribing information. Arrowhead Pharmaceuticals, Inc.; 2025.
- Watts GF, Rosenson RS, Hegele RA, et al. Plozasiran for managing persistent chylomicronemia and pancreatitis risk. New Engl J Med. 2025;392(2):127-137.
Indication & Important Safety Information
Indication
REDEMPLO® (plozasiran) is indicated as an adjunct to diet to reduce triglycerides in adults with familial chylomicronemia syndrome (FCS).
Important Safety Information
CONTRAINDICATIONS: None.
ADVERSE REACTIONS: Most common adverse reactions in REDEMPLO treated patients (incidence ≥10% of patients treated with REDEMPLO and > 5% more frequently than with placebo) are hyperglycemia, headache, nausea, and injection site reaction.